

About
The USACE has used bioaccumulation tests since the 1970s to make regulatory decisions regarding the management of dredged sediment. Prior to disposing of dredged sediment in ocean waters, the sediment must be evaluated to determine whether contaminants are present and whether the contaminant level may adversely impact the ecosystem at the ocean site. The USACE strives to protect human health and the environment by accurately identifying persistent, bioaccumulative, toxic substances and restricting disposal of these sediments. To help interpret data from these bioaccumulation tests, the USACE uses published BSAF data. To help meet the USACE need, the BSAF database was developed in the 1990s as a centralized repository of BSAF data. The database is a compilation of data, providing information on ratios between aquatic species and hydrophobic organic chemicals and organotin compounds. The source of data is primarily the peer-reviewed literature and reports submitted by U.S. government agencies. The database is updated annually with new records.
What is BSAF?
Biota-sediment accumulation factor (BSAF) is the ratio of the lipid-normalized concentration of a hydrophobic organic chemical (HOC) in an organism to the organic carbon-normalized concentration of the HOC in the sediment the organism was exposed to.
BSAF values are typically calculated using the following (or similar) formula:
![]()
![]() -the HOC concentration in the organism (wet or dry weight)
-the HOC concentration in the organism (wet or dry weight)
![]() -the lipid concentration (fraction) in the organism (wet or dry weight)
-the lipid concentration (fraction) in the organism (wet or dry weight)
![]() -the HOC concentration in the sediment (generally dry weight)
-the HOC concentration in the sediment (generally dry weight)
![]() -the total organic carbon content (fraction) of the sediment (generally dry weight)
-the total organic carbon content (fraction) of the sediment (generally dry weight)
HOCs preferentially partition to lipids so researchers commonly normalize concentrations of HOCs to lipid concentration in the tissue. This is accomplished by dividing tissue HOC concentration by lipid concentration to form lipid-normalized data. The concentration of total organic carbon (often referred to as TOC or OC) in sediment may also influence bioavailability because HOCs preferentially partition into the organic material in sediments because of the similar chemical nature of the organic material to the HOCs. Researchers normalize contaminants to sediment organic carbon by dividing the sediment contaminant concentration by the content of the sediment TOC.
In the past, sediment-based bioaccumulation factors were named accumulation factor, partitioning factor, bioavailability index and biota-sediment factor. All relate bioaccumulation to the concentration of a contaminant in the sediment. Due to inconsistency in naming, the term biota-sediment accumulation factor (BSAF) was proposed by Ankley et al. (1992). BSAFs have been used to both predict the amount of contaminant likely to be accumulated from sediment at equilibrium and for data comparison among species, compounds, sediment types, and contaminant concentrations.
Example
Example: Normalize the concentration of contaminant to lipid concentration in the tissue for an organism that has 308 ng PCB/g dry weight tissue with a lipid content of 8.4% dry weight (fraction= 0.084). Note the tissue concentration and lipid content must both be based on either wet weight or dry weight.
Lipid-normalized concentration:
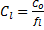
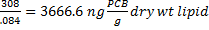
Example: Normalize the concentration of contaminants to sediment organic carbon for a sediment having 23 ng PCB/g dry weight and a mean TOC content of 2.8% TOC (fraction= 0.028)
Carbon-normalized concentration:
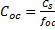
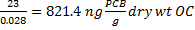
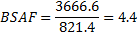
Structure
BSAF data is provided with limited supporting information (see Table 1 below). We recommend you revisit the literature to confirm your findings. We acknowledge there are other types of information that may be of value to your search that are not reported here. If you are interested in additional BSAF supporting information, please e-mail your ideas to the database manager and we will consider incorporating them going forward (E-mail database manager).
Data Quality Objectives
Systematic searches are conducted using a variety of resources including in-house and public databases. For each identified paper, an electronic copy is obtained for evaluation. Studies meeting the minimum data quality objectives listed below are initially accepted for further review:
- BSAF formula is defined as described above (i.e., BSAF is normalized).
- Exposure to a hydrophobic organic compound or organotin compound.
- Test species was a fish or other aquatic organism (e.g., mollusks, worms).
Upon further review papers must clearly identify the test substance, test species, and experimental conditions. Exclusion of a paper frequently occurs and is generally related to unclear or inexact details of the methods and results. Papers of interest to the BSAF include aquatic based laboratory or field investigations with a focus on fishes, worms, and bivalves as these are commonly used in bioaccumulation testing. Standard bioaccumulation tests are commonly conducted using Macoma nasuta (clam), and Alitta virens (polychaete) for marine tests, and Lumbriculus variegatus (an oligochaete) for freshwater tests. If the author reports the lipid-normalized concentration for the organism and the carbon-normalized concentrations for the sediment - or - provides the appropriate data (i.e., lipid and organic carbon content, tissue and sediment concentration) to do so, then ERDC will calculate BSAF.
Other supporting information is collected, but studies do not report the same level of detail. As a result, some data fields are not reported. No attempt is made to assign value to papers in the BSAF (e.g., Grade A, B, C; reliable or unreliable, etc.). We assume each user has a unique set of exclusion criteria to meet their needs. See the reference section below to learn more about selecting and using bioaccumulation data. The BSAF attempts to provide enough supporting information for you to begin selecting papers for a closer look. To understand the BSAF results, please consider obtaining a copy the original paper.
BSAF Data Counts
BSAF Data Fields and Descriptions
Table 1. Field descriptions for data presented online.
| Field Name | Description |
|---|---|
| BSAFC | BSAF calculated by ERDC (dependent on appropriate detail and availability of data). |
| BSAFR | BSAF reported in article. |
| CAS | CAS is the unique numerical identifier for the chemical assigned by the Chemical Abstract Service (CAS). |
| Chemical | Name of test chemical. |
| Chemical Spiked | Sediment spiked with chemical (Yes or No). Commonnly Yes in laboratory experiments, but commonly No in field studies. |
| DOI | The digital object identifier (DOI) that uniquely identifies an article and used to easily locate the article. |
| EXP duration | Length of laboratory experiment (e.g., 28 days). For field studies the entry is commonly not applicable (N/A). |
| GenusSpecies | Genus and species of test animal. |
| Lipid % | Lipid content. |
| Moisture | BSAF based on wet or dry -or- wet and dry weight (commonly tissue and lipid based on wet weight, while sediment and TOC based on dry weight). |
| Note | Can include information about the laboratory experiment such as whether animals were fed, purged, water exchange rates, loading ratio (e.g., TOC to animal), etc. |
| Reference | Unique identification number autogenerated by database for each reviewed article. |
| Sediment | Natural-collected from field; Artificial-made in laboratory; Amended-collected from field and amended w/other substance. Field studies use 'natural' sediment while laboratory studies sometimes use 'artificial' or 'amended'. |
| Sediment Locale | Where the sediment was collected from. |
| Source; Station | Where BSAF was found in the article (e.g., Table 1 or Figure 2). If BSAF is tied to a sample station this is also entered. |
| Study | Field Study, Laboratory Experiment, Mesocosm. |
| Subdivision, Country | Country and sudivision (UN/LOCODE CODE) where sediment was collected from. |
| Tissue | Tissue used to determine chemical concentration. |
| TOC % | Total organic carbon content. |
| ug/kg lipid | Lipid normalized concentration in organism. |
| ug/kg TOC | Carbon normalized concentration in sediment. |
References and other readings
Ankley, G. T., P. M. Cook, A. R. Carlson, D. J. Call, J. A. Swenson, H. F. Corcoran and R. A. Hoke. 1992. Bioaccumulation of PCBs from sediments by oligochaetes and fishes: comparison of laboratory and field studies. Canadian Journal of Fisheries and Aquatic Sciences 49:2080-2085. doi.org/10.1139/f92-231
[ASTM] American Society for Testing and Materials. 2010. Standard guide for determination of the bioaccumulation of sediment-associated contaminants by benthic invertebrates. E1688-10. ASTM annual book of standards, Vol 11.06. Biological effects and environmental fate; biotechnology; pesticides. West Conshohocken (PA): ASTM. 57 p.
Burkhard, L. P. 2003. Factors influencing the design of bioaccumulation factor and biota-sediment accumulation factor field studies. Environmental Toxicology and Chemistry: An International Journal 22:351-360. doi.org/10.1002/etc.5620220216
Burkhard, L. P., P. M. Cook and M. T. Lukasewycz. 2005. Comparison of biota-sediment accumulation factors across ecosystems. Environmental Science and Technology 39:5716-5721. doi.org/10.1021/es050308w
Burkhard, L. P., J. A. Arnot, M. R. Embry, K. J. Farley, R. A. Hoke, M. Kitano, H. A. Leslie, G. R. Lotufo, T. F. Parkerton, K. G. Sappington, G. T. Tomy, K. B. Woodburn. 2012. Comparing laboratory-and field-measured biota-sediment accumulation factors. Integrated Environmental Assessment and Management 8:32-41. doi.org/10.1002/ieam.218
Judd, N., L. Tear and J. Toll. 2013. From sediment to tissue and tissue to sediment: an evaluation of statistical bioaccumulation models. Integrated Environmental Assessment and Management 10:102-113. doi.org/10.1002/ieam.1484
Klimisch, H. J., M. Andreae and U. Tillman. 1997. A systematic approach for evaluating the quality of experimental toxicology and ecotoxicology data. Regulatory Toxicology and Pharmacology 25: 1-5. doi.org/10.1006/rtph.1996.1076
Lotufo, G.R., J. M. Biedenbach, J. D. Farrar, M. K. Chanov, B. W. Hester, C. R. Warbritton, J. A. Steevens, J. M. Netchaev, A. J. Bednar, and D. W. Moore. 2022. Interlaboratory comparison of three sediment bioaccumulation tests. Environmental Toxicology and Chemistry 41:1260-1275. doi.org/10.1002/etc.5300
[OECD] Organisation for Economic Co-operation and Development. 2008. OECD guidelines for the testing of chemicals, TG 315. Bioaccumulation in sedimentdwelling benthic oligochaetes. Paris (FR): OECD.
Parkerton, T. F., J. A. Arnot, A. V. Weisbord, C. Russom, R. A. Hoke, K. Woodburn, T. Traas, M. Bonnell, L. P. Burkhard and M. A. Lampi. 2008. Guidance for evaluating in vivo fish bioaccumulation data. Integrated Environmental Assessment and Management 4:139-155. doi.org/10.1897/IEAM_2007-057.1
[USEPA] US Environmental Protection Agency. 2000. Methods for measuring the toxicity and bioaccumulation of sediment-associated contaminants with freshwater invertebrates. 2nd edition. EPA 600/R-99/064. Duluth (MN): Office of Research and Development, Mid-Continent Ecology Division, and Washington (DC): Office of Water, Office of Science and Technology, USEPA.
[USEPA] US Environmental Protection Agency. 2009. Methodology for deriving ambient water quality criteria for the protection of human health (2000) Technical Support Document. Volume 3: Development of site-specific bioaccumulation factors. EPA-822-R-09-008. Washington (DC): Office of Water, Office of Science and Technology, USEPA
[USEPA/USACE] US Environmental Protection Agency/US Army Corps of Engineers. 1998. Evaluation of dredged material proposed for discharge in waters of the U.S. - Testing manual inland testing manual. EPA-823-B-98-O04. Washington (DC): Office of Water, Office of Science and Technology, and Operation, Construction, and Readiness Division, USEPA and Department of the Army, USACE.
Van Geest, J. L., D. G. Poirier, P. K. Sibley and K. R. Solomon. 2010. Measuring bioaccumulation of contaminants from field-collected sediment in freshwater organisms: a critical review of laboratory methods. Environmental Toxicology and Chemistry 29:2391-2401. doi.org/10.1002/etc.326
Weisbrod, A. V. and 13 co-authors. 2007. Workgroup report: review of fish bioaccumulation databases used to identify persistent, bioaccumulative, toxic substances. Environmental Health Perspectives 115:255-261. doi.org/10.1289/ehp.9424